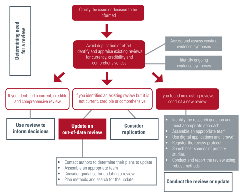
Resources for researchers
The COVID-19 pandemic has led to an explosion of activities among all types of researchers, including in the evidence-synthesis, technology-assessment and guideline-development communities. COVID-END has assembled resources for individual researchers and research teams who are involved or who want to become involved in preparing timely, relevant and high-quality evidence syntheses and guidelines to support decision-making about COVID-19:
- priorities for new syntheses and guidelines
- supports for evidence synthesizers
- supports for guidance developers
There are many different types of evidence synthesis and the resources provided supports for evidence synthesizers focus on: rapid reviews, scoping reviews, systematic reviews (SR), and living SRs. To learn more, see our definitions of key types of evidence synthesis.
Relevant reading:
Akl EA, Haddaway NR, Rada G, Lotfi T. Evidence synthesis 2.0: When systematic, scoping, rapid, living, and overviews of reviews come together. Journal of Clinical Epidemiology 2020; 123: 162-165 https://doi.org/10.1016/j.jclinepi.2020.01.025
Gough D, Thomas J, Oliver S (2019). Clarifying differences between reviews within evidence ecosystems. Systematic Reviews 2019; 8 (1): 170. https://systematicreviewsjournal.biomedcentral.com/articles/10.1186/s13643-019-1089-2
To share any comments or to suggest further resources to consider adding, please contact covid-end@mcmaster.ca.
Download a PDF of 'Resources for researchers considering and conducting COVID-19 evidence syntheses'
View the interactive flow diagram